

Everything you need to know about China’s Patent Term Extension (PTE)
By Hongyue Li | Posted on March 10, 2026

The pharmaceutical field is one that relies most heavily on patent protection. The development of new drugs involves high costs, long cycles, and significant risks. Without patent protection, new drug R&D enterprises would find it difficult to recoup R&D costs and earn reasonable profits, thereby reducing their incentive to develop new drugs. Meanwhile, due to the special nature of pharmaceuticals, countries worldwide subject drugs to stringent administrative regulation. Drug marketing requires rigorous approval from drug regulatory authorities, including requirements for non-clinical safety evaluations and clinical trials. This means that even after a drug invention obtains a patent grant, it cannot be implemented for a considerable period before receiving marketing authorisation, thereby objectively shortening the effective patent term for pharmaceutical patents.
To compensate for the time occupied by the review and approval process for new drug marketing, the China National Intellectual Property Administration (CNIPA) introduced the Patent Term Extension (PTE) provision for pharmaceutical invention patents for the first time in the Fourth Amendment of the Chinese Patent Law. This amended Patent Law came into effect on June 1, 2021. The relevant supporting regulations—the Implementing Regulations of the Patent Law and the Patent Examination Guidelines—were revised and also came into effect on January 20, 2024. Since then, the implementation and examination of PTE have entered the practical stage. In this article, an overview and operational practices for PTE in China are provided by answering frequently asked questions.
1. Which provision in the Chinese Patent Law pertains to PTE for pharmaceutical invention patents? What is its specific content?
Article 42, paragraph 3 of the Patent Law stipulates: To compensate for the time occupied by the review and approval process for new drug marketing, for invention patents related to new drugs that have obtained marketing authorisation in China, the patent administration department under the State Council (e.g. CNIPA) shall, upon request by the patentee, grant a patent term extension. The extension period shall not exceed five years, and the total effective patent term after the new drug is approved for marketing shall not exceed fourteen years.
2. How are the terms “new drug” and “invention patent related to a new drug” mentioned in the provision interpreted?
According to the Patent Law and its Implementing Regulations, only a “new drug that meets stipulations” can serve as the basis for a pharmaceutical patent term extension.
Specifically, the “new drug” in the provision refers to:
Innovative Drug (Category 1): Innovative drugs not yet marketed domestically or internationally.
Improved New Drugs of the following five categories:
- Chemical Drug Category 2.1: Drugs involving esterification or salification of a known active ingredient.
- Chemical Drug Category 2.4: Drugs containing a known active ingredient for a new indication.
- Preventive Biological Product Category 2.2: Vaccines with improvements to the vaccine strain.
- Therapeutic Biological Product Category 2.2: Biological products with a new indication.
- Traditional Chinese Medicine (TCM) Category 2.3: TCM with added functions/indications.
The “invention patent related to a new drug” in the provision refers to a patent for the active ingredient of a new drug that is approved for marketing for the first time by the drug regulatory department under the State Council (e.g., the NMPA).
For innovative drugs and qualifying improved new drugs approved for marketing by the NMPA, a patent term extension may be granted for product invention patents, preparation process invention patents, or medical use invention patents related to the drug’s active substance. The new drug active substance or active ingredient typically refers to “the drug active substance or ingredient that plays a substantive role in preventing, treating, or diagnosing a disease within a new drug”.
Examples of determining whether an invention patent is related to a new drug:
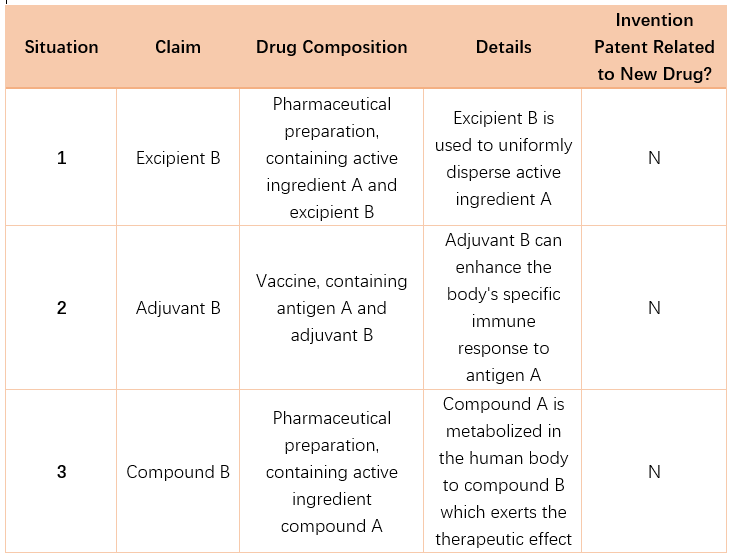
3. What are the conditions for PTE?
The conditions for requesting a pharmaceutical patent term extension include:
a) The publication date of the patent grant for which compensation is requested shall be earlier than the date the drug marketing authorisation application is approved;
b) At the time of submitting the compensation request, the patent right must be valid.
c) The patent has not previously received a pharmaceutical patent term extension.
d) The claims of the patent for which compensation is requested include the technical solution related to the new drug that has obtained marketing authorisation.
e) If multiple patents exist for the same drug, the patentee can only request a patent term extension for one of these patents.
f) If a patent covers multiple drugs, a patent term extension request for that patent can be made only for one drug.
4. How to interpret “one drug” in conditions (e) and (f) above?
Different strengths of the same drug belong to “one drug”.
Drugs with different indications belong to different classes of drugs.
Examples:
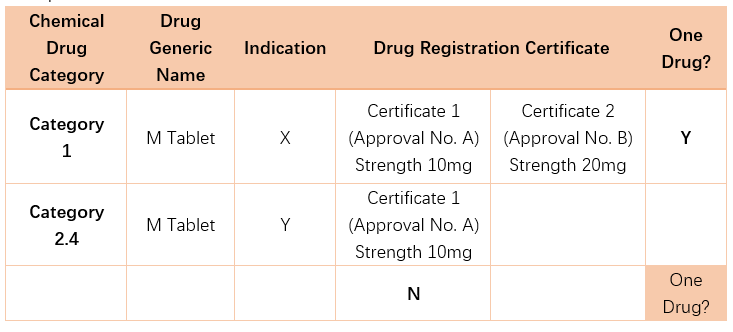
5. Who is the eligible party to request PTE?
The patentee shall submit the request for a pharmaceutical patent term extension.
If the patentee is not the same as the drug marketing authorisation holder, when submitting a compensation request, the patentee shall obtain written consent from the drug marketing authorisation holder. This is to resolve potential disputes between the parties as early as possible.
6. Are there timing requirements for submitting a PTE request?
The patentee shall submit the request to the Patent Office within three months of the date on which the drug marketing authorisation application is approved and pay the corresponding fees.
If marketing authorisation for different strengths of the same drug is approved on different dates, the three months are calculated from the earliest approval date.
For drugs granted conditional marketing authorisation, the request shall be submitted to the Patent Office within three months of the date formal marketing authorisation is obtained in China. Still, the extension period is calculated from the date the conditional marketing authorisation was obtained.
7. What materials does the patentee need to submit when requesting PTE?
When submitting a request for pharmaceutical patent term extension, the patentee shall submit a request form, the drug registration certificate (including the package insert), a comparison table between the technical solution related to the new drug and the specified claims, and necessary supporting documents (e.g., as applicable, production process, quality standards, relevant sections from the “M4: Common Technical Document for Registration of Pharmaceuticals for Human Use (CTD)” filed with the NMPA, information disclosed in the WHO International Nonproprietary Name (INN) list, etc.).
- The request form shall state drug marketing authorisation-related information, including drug name (generic name), drug approval number, drug registration category, approved indication, marketing authorisation date, and marketing authorisation holder, etc.
- State the invention patent information for which the term extension is requested: including patent number, title of invention, and patentee, etc.
- Specify the claim(s) related to the new drug that has obtained marketing authorization, and explain the reasons for compensation with supporting documents: Specifically explain the rationale that the specified claim(s) include the technical solution related to the new drug, the calculation basis for the requested extension period, etc., and clarify the technical solution protected during the pharmaceutical patent term extension period.
When submitting the request, the patentee shall provide supporting documents, including:
- Drug registration certificate and its attachments (e.g., package insert), etc.
- If the patentee and the drug marketing authorisation holder are not the same, the patentee shall submit written consent from the drug marketing authorisation holder and other materials.
- Technical materials are used to explain that the specified claim(s) include the technical solution related to the new drug and to determine the scope of patent protection during the pharmaceutical patent term extension period, etc.
- Other relevant supporting documents as required by the Patent Office.
8. How to understand “technical solution related to the new drug”?
The technical solution related to the new drug shall be based on the structure, composition and content, approved production process, and indication of the new drug, as approved by the drug regulatory department under the State Council (e.g., NMPA).
- Generally, the subject matter type of the technical solution related to the new drug should be consistent with the subject matter type of the specified claim(s), and it should be a technical solution related to the active substance of the approved new drug.
- The active substance of a new drug usually refers to the drug component that plays a substantive role in preventing, treating, or diagnosing the disease within the new drug.
- The approved indication refers to the indication for which the involved drug is approved at this time.
Example:
Background: The newly approved drug belongs to Chemical Drug Category 2.1, with active substance A1, and its approved indication is for the treatment of asthma.
If the specified patent claim 1 claims compound A with a general formula, then the corresponding technical solution related to the new drug is considered to be compound A1.
If the specified patent claim 2 claims the use of compound A with a general formula in the manufacture of a medicament for treating asthma, then the corresponding technical solution related to the new drug is considered to be the use of compound A1 in the manufacture of a medicament for treating asthma.
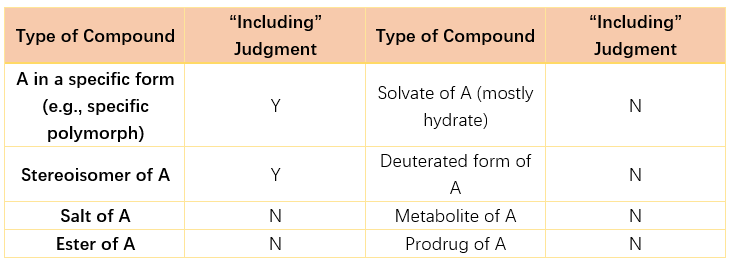
9. How to determine whether the specified claim(s) include the technical solution related to the new drug that has obtained marketing authorisation?
Several scenarios for “include” are as follows:
- The technical features of the specified claim(s) and the technical solution related to the new drug are identically expressed;
- Although there are differences in the expression of technical features between the specified claim(s) and the technical solution related to the new drug, they are merely simple textual transformations, and their substantive meanings are the same.
- One or more technical features of the specified claim(s) are broader concepts (superordinate concepts) of the relevant technical features of the technical solution related to the new drug;
- The difference between the technical solution related to the new drug and the specified claim(s) is only that the numerical feature(s) in the new drug technical solution are specific values or sub-ranges within the corresponding numerical range feature(s) of the specified claim.
Example 1:
Background: The newly approved drug belongs to Chemical Drug Category 2.1, with active substance A1, and the approved indication is for the treatment of asthma.
The specified patent claim 1 claims compound A of a general formula. If, upon examination, the general formula A is deemed to include A1, then it can be determined that the specified claim 1 includes the technical solution related to the new drug that has obtained marketing authorisation.
Example 2:
Specified claim: A compound A, characterised by having structure B.
Example 3:
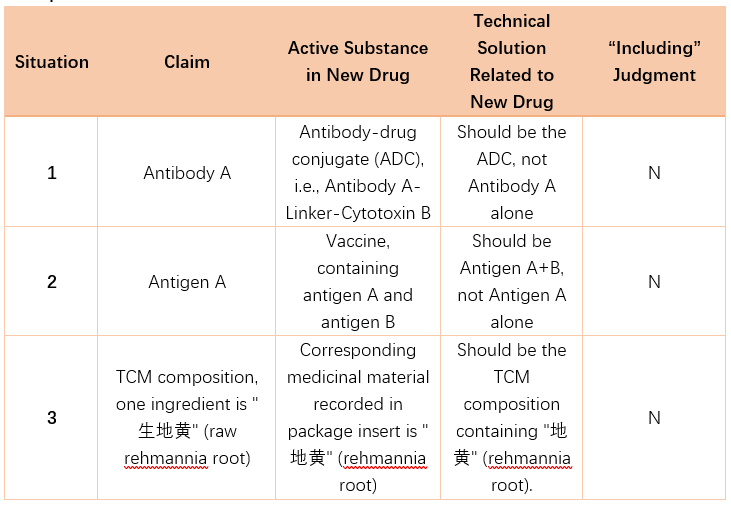
10. How is the pharmaceutical patent term extension period calculated?
The formula for calculating the pharmaceutical patent term extension period is:
Pharmaceutical Patent Term Extension Period = China Drug Marketing Authorisation Date – Patent Filing Date – 5 years (≤ 5 years)
Total Effective Patent Term = 20-year Expiry Date – China Drug Marketing Authorization Date + PTA + PTE (≤ 14 years)
The pharmaceutical patent term extension period has dual limitations: first, the extension period shall not exceed 5 years; second, the total effective patent term after the new drug is approved for marketing shall not exceed 14 years.
11. What are the special scenarios in the calculation of the pharmaceutical patent term extension period?
Several scenarios are as follows:
- If “one drug” has multiple drug approval numbers (e.g., different strengths) with different marketing approval dates, the extension period shall be calculated based on the earliest marketing approval date.
- If the drug registration certificate is corrected, the extension period shall be calculated based on the “original approval date” stated in the remarks column of the corrected drug registration certificate.
- For drugs granted conditional marketing authorisation, the extension period shall be calculated from the date conditional marketing authorisation was obtained, not from the date formal marketing authorisation was obtained.
12. What is the scope of patent protection during the pharmaceutical patent term extension period?
During the pharmaceutical patent term extension period, the scope of protection of the patent is limited to the specific new drug approved for marketing by the National Medical Products Administration (NMPA) and to the technical solution related to the approved indication of that new drug.
Example 1:
Background: The newly approved drug belongs to Chemical Drug Category 2.1, with active substance A1, and the approved indication is for the treatment of asthma.
The specified patent claim 1 states that compound A has the general formula. If, upon examination, the general formula A is deemed to include A1, then it can be determined that the specified claim 1 includes the technical solution related to the new drug that has obtained marketing authorisation.
In this scenario, during the pharmaceutical patent term extension period, the protection scope of the patent is limited to “Compound A1 for treating asthma”, not the patent claim 1.
Example 2:
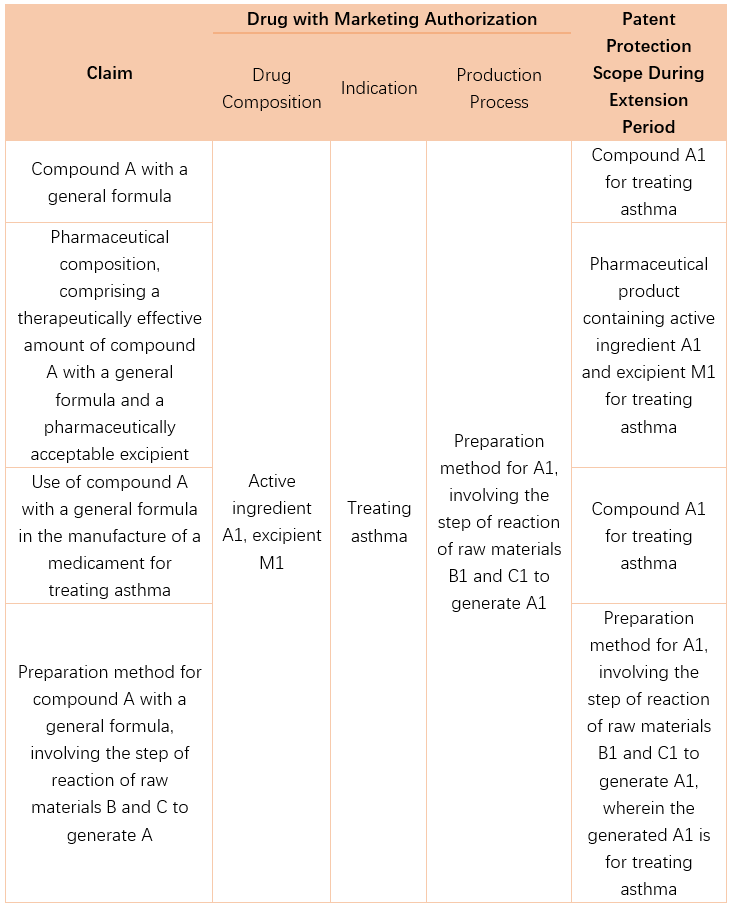
13. If there are defects in the submitted pharmaceutical patent term extension request, is there an opportunity for correction?
If, upon examination, the request does not meet the compensation conditions, the CNIPA may issue a Notification to Make Rectification or an Office Action on Pharmaceutical Patent Term Extension. The requester/ patentee may submit amendments and/or observations in their response.
14. Are there any remedies if one disagrees with a PTE decision made by the CNIPA?
If one disagrees with the PTE decision, one may apply for administrative reconsideration with the CNIPA or initiate administrative litigation.
The patent term extension (PTE) system is a critical initiative for aligning China’s pharmaceutical intellectual property protection framework with international standards. It reshapes the value assessment system for innovative drugs. For pharmaceutical companies, PTE extends the market exclusivity period for core innovative products and prolongs the value realisation cycle of patent assets. Accordingly, companies can proactively adjust their intellectual property strategies: fully integrate China into globally synchronised R&D and submission systems to ensure eligibility for PTE from the early stages of development; align patent portfolios with global launch timelines, planning application pathways for core patents; and foster high-level integration of R&D, regulatory affairs, and intellectual property management at the decision-making level. This approach enables genuine competition through the generation of innovation and a strategic global vision.
Filed under: Insights
